Clinical Trials - Targeted Solutions:
- Rapid Trial Activation
- Assessment & Roadmap Development
- Clinical Trials Office/Research Operations Office
Research - Targeted Solutions:
Clinical Trials - Targeted Solutions:
- Rapid Trial Activation
- Assessment & Roadmap Development
- Clinical Trials Office/Research Operations Office
Research - Targeted Solutions:
Clinical Operations
Site Identification, Selection, Activation and Ongoing Support
Vitalief’s Site Identification, Selection, Activation, and Ongoing Support services give Sponsors and CROs a complete solution to accelerate trial start-up and sustain consistent execution across all sites, regardless of phase, indication, or therapeutic area. Our specialized team of seasoned site experts serves as the central point of communication and coordination, managing every step from feasibility through trial execution. This includes site identification and qualification, oversight of the selection process, regulatory and IRB submissions/approvals, budget negotiations, contract execution, pharmacy preparation, and protocol-specific training. We also schedule and conduct Site Initiation Visits, ensuring every site receives consistent guidance, messaging, and compliance oversight.
But activation is only the beginning. Our ongoing site support keeps sites engaged and equipped throughout the trial lifecycle. We provide proactive communication, operational oversight, and troubleshooting to minimize disruptions, reduce mid-trial risks, and keep enrollment on track. By maintaining alignment between Sponsors, CROs, and Sites, we ensure that studies stay compliant, efficient, and milestone-ready.
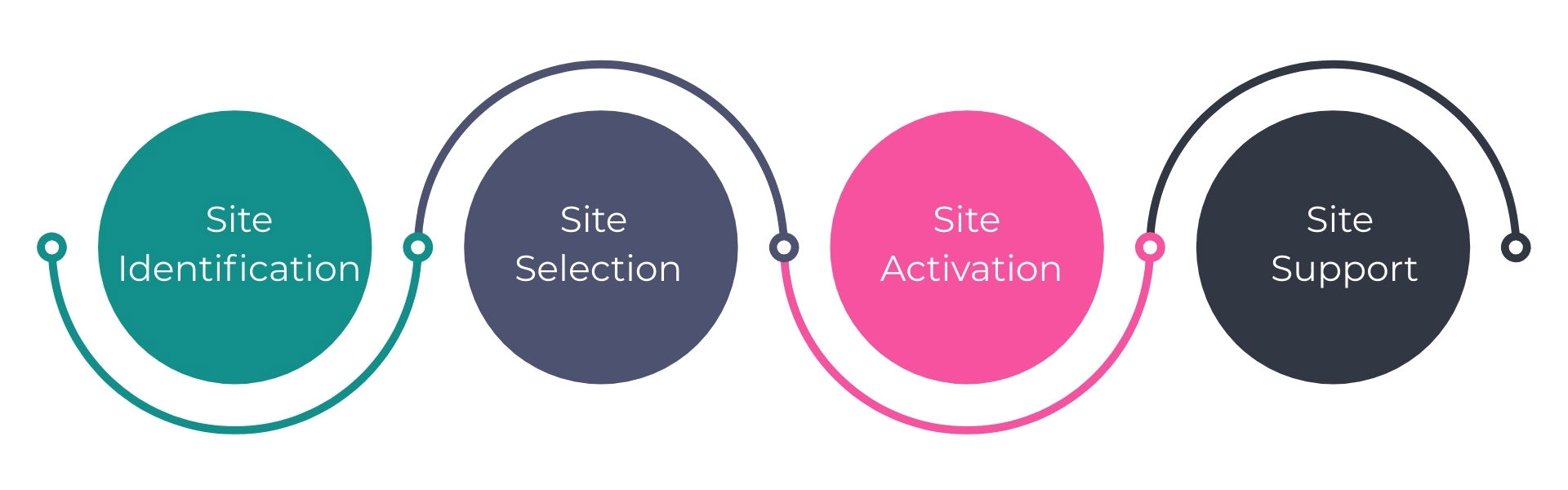
For Sponsors and CROs, this means shorter timelines to “first patient in,” smoother enrollment, and greater confidence in hitting trial milestones. For Sites, it means a streamlined onboarding process, reduced administrative burden, and expert support from activation through closeout—ensuring each trial is launched and sustained with the right infrastructure in place.
Why Choose Vitalief
Sponsors, CROs, and Sites choose Vitalief’s Clinical Operations services because we provide hands-on, scalable support that drives efficiency, quality, and compliance from trial startup through closeout. Our experienced teams and tailored solutions help align all stakeholders, reduce delays, and ensure consistent execution across trials of all sizes and complexities.
Let's get started!
© 2021-2025 Vitalief. All rights reserved.
Vitalief • New Brunswick, NJ 08901 • United States
By using the website you accept our privacy policy. Choose your cookie consent:

